release time:2021-07-26 15:11:59
Common in vitro diagnostic devices:
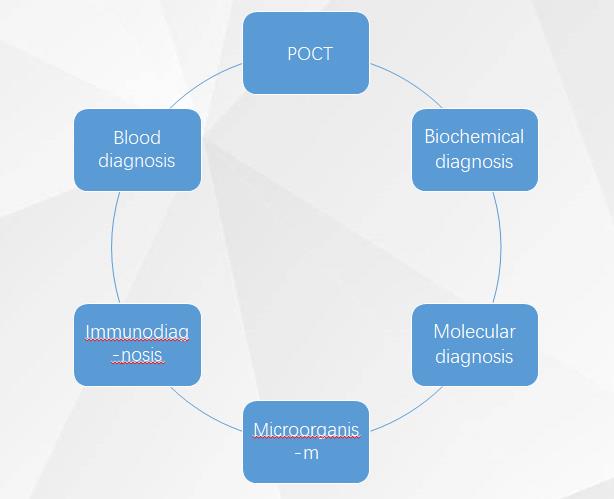
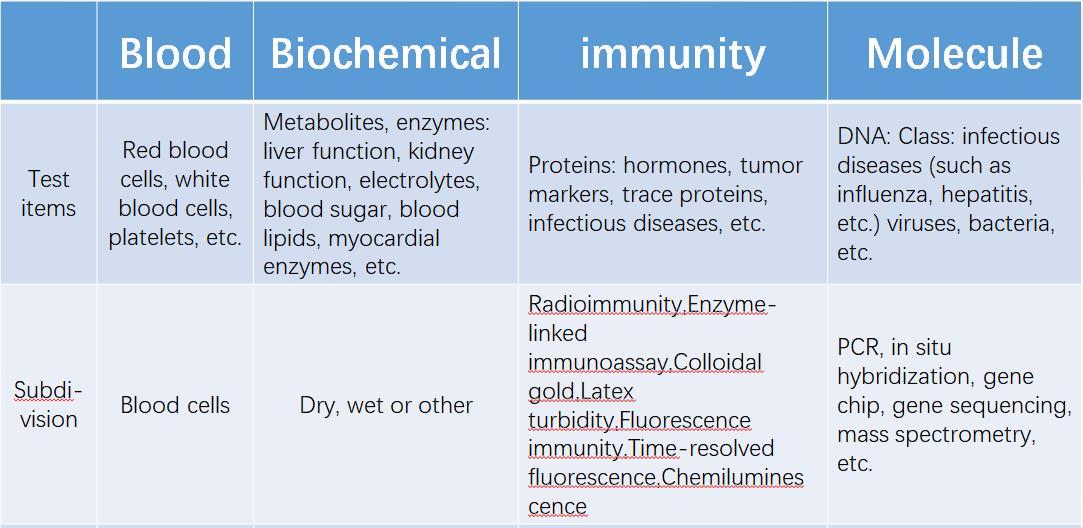
According to the detection category, it can be divided into blood test, biochemical diagnosis, immunodiagnosis, molecular diagnosis, microbiological diagnosis, POCT, etc.
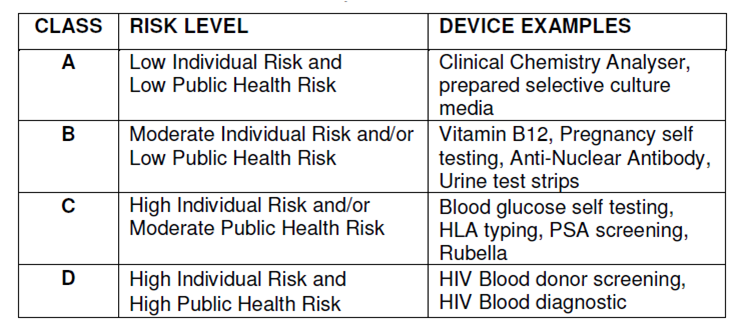
The FDA classifies medical devices (including IVD products) into Class I, Class II, or Class III based on the level of regulatory control required to reasonably ensure safety and effectiveness. The classification of IVD (or other medical devices) determines the appropriate pre-marketing process.

2022-03-29
The main purpose of animal testing of medical devices is to study the safety of medical devices. Often the subject of animal testing in medical research may be a new diagnostic method and principle, or the mechanism of action of a new drug, etc.
2022-02-25
In biochemical tests, anticoagulants can be selected to inhibit certain clotting factors in the blood and prevent blood from clotting. In whole blood or plasma tests, a different anticoagulant is usually needed to keep the collected venous blood from coagulating.
2021-12-27
Routine blood tests, blood biochemical tests, fecal tests, urine tests, skin tests, etc. are the main items of laboratory tests in animal hospitals at present.